Prof. Xinhe Bao’s research team in the State Key Laboratory of Catalysis has recently made research progress in metal-oxide interface enhanced CO2 electroreduction. The results are published in Journal of the American Chemical Society (J. Am. Chem. Soc. 2017, 139, 5652).
The electrochemical CO2 reduction reaction (CO2RR), powered by renewable electricity or surplus nuclear electricity, can be used to directly prepare carbon monoxide, formic acid, hydrocarbon, alcohol, other high value fuel and chemicals under relatively mild reaction conditions in one step. The process can simultaneously achieve efficient CO2 conversion and clean energy storage. At present, designing efficient catalysts to decrease the overpotential and enhance reaction selectivity is the challenging research focus for CO2RR.
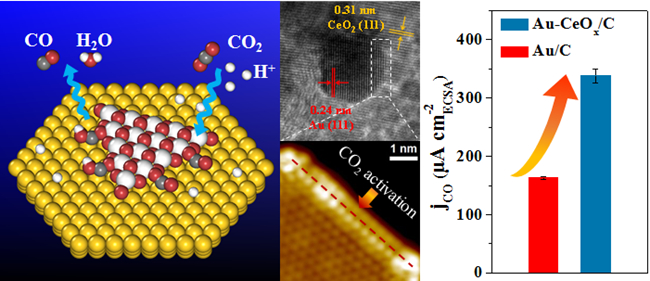
Based on the previous CO2RR studies and the understanding of the metal-oxide interfacial confinement catalysis, the research team designs and prepares carbon supported Au-CeOx catalyst with metal-oxide interface structure, and investigates the relationship between the Au-CeOx interface and catalytic performance of CO2RR. The CO Faradaic efficiency reaches 89.1% over Au-CeOx/C at −0.89 V vs. reversible hydrogen electrode (RHE), which is significantly higher than 59.0% and 9.8% over Au/C and CeOx/C at the same potential. The CO geometric current density over Au-CeOx/C (12.9 mA cm−2) is about 1.6 times of that over Au/C (8.3 mA cm−2) at −0.89 V vs. RHE. In situ scanning tunnelling microscopy and synchrotron-radiation photoemission spectroscopy show that the Au-CeOx interface is dominant in enhancing CO2 adsorption and activation, which can be further promoted by the presence of hydroxyl groups. Density functional theory calculations indicate that the Au-CeOx interface is the active site for CO2 activation and the reduction to CO, where the synergy between Au and CeOx promotes the stability of key carboxyl intermediate (*COOH) and thus facilitates CO2RR. Similar interface-enhanced CO2RR is further observed on Ag-CeOx, demonstrating the generality of the strategy for enhancing CO2RR. The research results provide a new way to regulate the CO2RR performance, and enrich and expand the concept of nano-confined catalysis proposed by the research team.
This work was financially supported by Natural Science Foundation of China, National Key R&D Program of China and Strategic Priority Research Program of the Chinese Academy of Sciences. (By Dunfeng Gao, Yi Zhang, Zhiwen Zhou)