Prof. Xinhe Bao and Prof. Guoxiong Wang’s research team in the State Key Laboratory of Catalysis has recently made research progress in high-temperature electrocatalytic reduction of CO2. The results are published in Nano Energy (Nano Energy, 2018,50,43-51).
High-temperature CO2 electrolysis using solid oxide electrolysis cells (SOECs) is capable of reducing CO2 and water to syngas, hydrocarbon fuel at the cathode, and producing pure oxygen at the anode. SOECs have the advantages of possessing solid and modular structure, high energy efficiency and low cost. Therefore, SOECs have potential applications in CO2 conversion and surplus sustainable electricity storage. Conventional SOECs using Ni-YSZ cathode have demonstrated excellent electrochemical performance in CO2 electrolysis. However, the electrocatalytic performance of the Ni-YSZ cathode diminishes with time on stream due to carbon deposition and coarsening of Ni at high temperature in CO2 atmosphere. The research and development of novel perovskite or fluorite oxides as cathode materials to achieve efficient and stable CO2 electrolysis is a hot topic of SOECs in recent years.
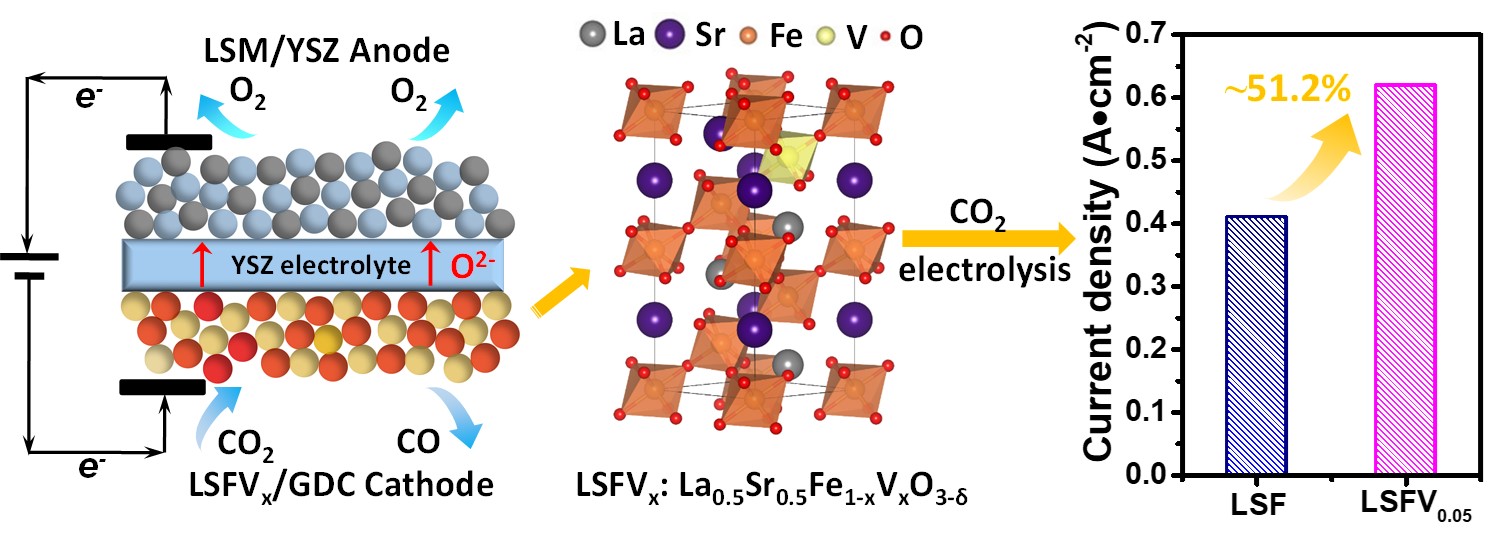
In this work, the researchers investigated the nanocomposites of La0.5Sr0.5Fe1-xVxO3-δ (LSFVx, x = 0, 0.05, 0.10, 0.15) with Ce0.8Gd0.2O1.9 (LSFVx/GDC) as cathode materials for pure CO2 electrolysis in SOEC. A higher valence V5+ is selected as the dopant for partial substitution of Fe3+ in La0.5Sr0.5FeO3-δ to tailor its oxygen vacancies and electronic structures. The introduction of vanadium promotes the formation of oxygen deficiencies in LSFVx/GDC, and alters the electronic structure of Fe, thus greatly enhances CO2 adsorption and dissociation. The LSFV0.05/GDC possesses the highest oxygen vacancy concentration at 800 °C as well as the highest CO2 adsorption and dissociation ability. The highest current density of 0.62 A cm-2 could be achieved over the LSFV0.05/GDC cathode at 1.6 V, which is about 51.2% increase compared with that over the LSF/GDC cathode. This study regulates oxygen vacancy concentration and CO2 activation/adsorption ability of SOEC cathode materials via doping metal element, which provides a new strategy to improve the performance of electrocatalytic CO2 reduction at the SOEC cathode.
This work was financially supported by Natural Science Foundation of China, National Key R&D Program of China, Dalian Institute of Chemical Physics and Strategic Priority Research Program of the Chinese Academy of Sciences. (By Yingjie Zhou and Zhiwen Zhou)