Recently, Associate Researcher Dr. Yuefeng Song et al. from the team of Carbon-Based Resource Electrocatalysis Research of the National Key Laboratory of Energy Catalysis Conversion at Dalian Institute of Chemical Physics, CAS, in collaboration with Prof. Guoxiong Wang’s team at Fudan University and Academician Meilin Liu’s group at the Georgia Institute of Technology, have made significant progress in elucidating the mechanisms underlying the high-temperature oxygen evolution reaction (OER) at the anode of solid oxide electrolysis cells (SOECs).This study focuses on the effect of A-site cation ordering in perovskite anodes on high-temperature OER performance, and systematically reveals how the order–disorder transition in the PrxBa2-xCo2O5+δ system influences the material’s microstructure and electrocatalytic activity.
SOECs are considered among the most promising technologies for CO2 reduction and energy conversion, owing to their high current density, excellent Faradaic efficiency, and low overpotential. Perovskite oxides are widely employed as anode materials in SOECs; however, the influence of A-site cation ordering on the kinetics of the oxygen evolution reaction (OER) remains poorly understood. In this work, two perovskite anodes with different Pr contents - PrBaCo2O5+δ (PBCO-1.0) and Pr1.5Ba0.5Co2O5+δ (PBCO-1.5)- were synthesized. A combination of advanced characterization techniques, including synchrotron X-ray powder diffraction (SXRPD), high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM), X-ray absorption spectroscopy (XAS), and 18O isotope exchange experiments, was employed to systematically investigate the effect of A-site cation disorder on the electronic structure and high-temperature OER kinetics.
The study reveals that as the Pr content increases from 1.0 to 1.5, the crystal structure of PBCO undergoes a transition from an ordered tetragonal phase (P4/mmm) to a disordered orthorhombic phase (Pnma). This structural transformation disrupts the local symmetry of the Co-O coordination environment, enhances the orbital hybridization between Co 3d and O 2p states, and thereby improves oxygen ion mobility and accelerates surface oxygen exchange kinetics. Under operating conditions of 800 °C and 1.6 V, the PBCO-1.5 anode achieves a current density of 2.29 A cm-2, demonstrating outstanding high-temperature OER activity and stability.
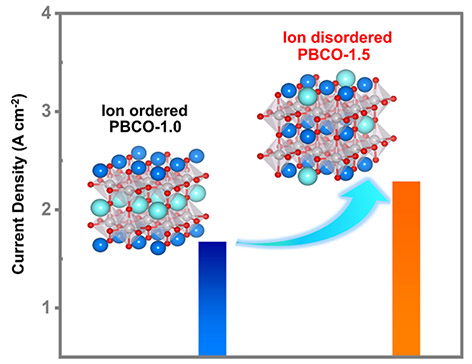
This study elucidates, through both experimental and theoretical investigations, the regulatory role of A-site cation ordering in perovskite oxides on the reaction pathway and kinetics of high-temperature OER. The findings offer valuable guidance for the rational design of high-performance anode materials for SOECs. The related research, entitled “Breaking the Ion Ordering in Perovskite Anode for Enhanced High-Temperature Oxygen Evolution Reaction Activity,” was published in the internationally renowned journal Journal of the American Chemical Society. The paper was co-first-authored by Dr. Lina Yu, a postdoctoral researcher from Group 523, and Xueyu Hu, a Ph.D. student at the Georgia Institute of Technology. This work was supported by the National Key R&D Program of China, the National Natural Science Foundation of China, and other funding agencies. (Written and illustrated by Lina Yu)
Article link: https://doi.org/10.1021/jacs.5c09331